Di Water Conductivity Vs Resistivity
It is pure water.
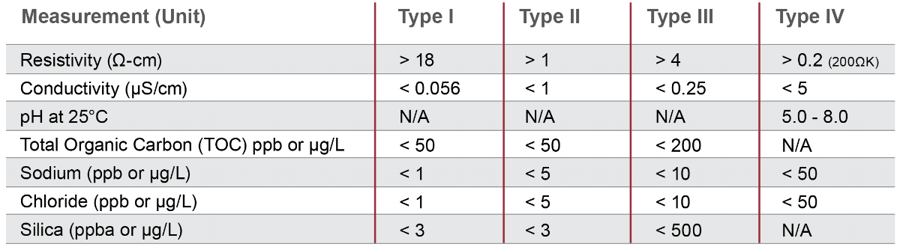
Di water conductivity vs resistivity. If the temperature rises by 1 c the conductivity of mains water will increase by approximately 2 whereas that of ultra pure water will increase by up to 6. Images may or may not be of the actual product. The de ionized water increases in quality with lower conductivities though higher grade de ionized di water will be more expensive and will take more time and energy to produce. Those involved in the production of semiconductor grade high purity water use resistivity units i e 18 2 mω cm.
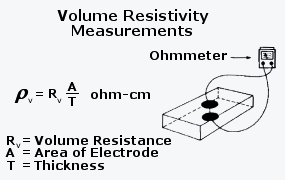
Pure water will not conduct electricity the rating is in meg ohms. Di or de ionized water is simply water that has had most of the mineral content and dissolved ion content removed. In conclusion the proper conductivity range for di water is typically 5 10 6 s m. 2 resistivity and conductivity are identical measurements differing only in the units of expression.
That afore mentioned range corresponds to 1 to 0 1 megohm cm. Both cost and suitability are factors are important. It is standard practise to correct conductivity and resistivity to 25 c a process which is carried out automatically by modern conductivity meters. The resistivity is the reciprocal of the conductivity.
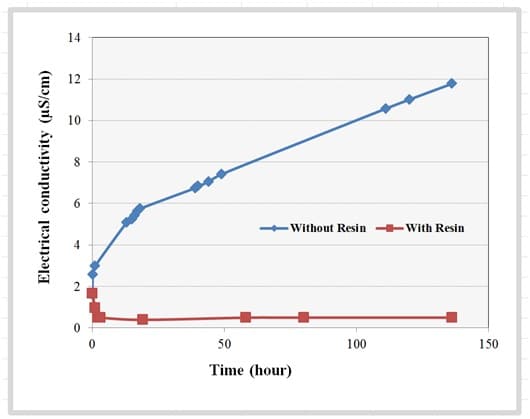
Deionized water di water diw or de ionized water often synonymous with demineralized water dm water is water that has had almost all of its mineral ions removed such as cations like sodium calcium iron and copper and anions such as chloride and sulfate deionization is a chemical process that uses specially manufactured ion exchange resins which exchange hydrogen and hydroxide ions. Power plant chemists and the pharmaceutical industry tend to prefer conductivity units such as µs cm or µmho cm. For defluxing cleaning applications the recommended di water quality should be 1 to 10 microsiemens cm. Any conductivity that is higher than 10 microsiemens cm is not recommended.
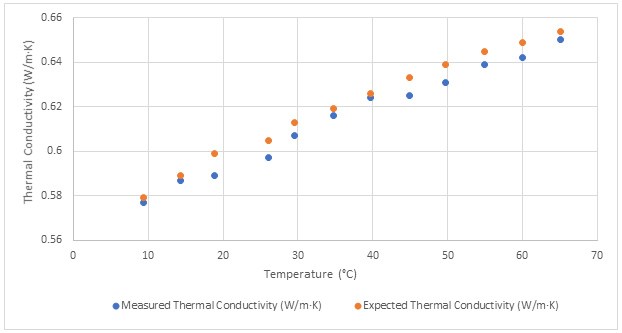
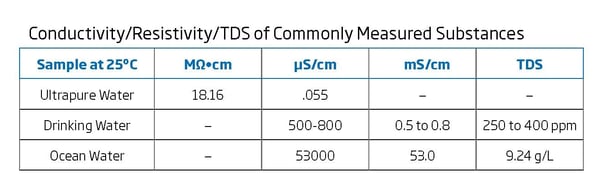
Conductivity vs resistivity vs ppm quick chart all readings are at 1 cm probe distance and a 25c temperature. High purity water resistivity conductivity measurement ultra pure water without any chemical impurities will still have a conductivity because of the presence of h and oh ions due to the self ionization of water. Resistivity is the reciprocal of conductivity and either may be used to inexpensively monitor the ionic purity of water. Most laboratory water purification systems contain a resistivity or conductivity meter and cell to monitor the purity level of the water.
Great for lab water pretreatment or great drinking water images are representative of the products. Conductivity and resistivity are reciprocal values. Resistivity is the reciprocal of conductivity and either may be used to inexpensively monitor the ionic purity of water. 800 460 9011 toll free.
The cleanliness of the water is rated as its resistance value.