Distilled Vs Deionized Water Ph

Deionized di water is water that has been treated to remove all ions typically that means all of the dissolved mineral salts.
Distilled vs deionized water ph. Ph electrodes will not give accurate ph values in distilled or deionized water because distilled and deionized water do not have enough ions present for the electrode to function properly. The basics of ph. However there is still much confusion about its significance in high purity water applications. The relationship between ph and deionized water.
What is distilled water. In this article you can find the explanation to understand the differences and similarities of these two different water. It s fine to use this type of leftover deionized water in place of distilled water. Industries and products that use deionized water.
You don t generally want to substitute one type of water for the other but if you have deionized water made from distilled water that has been sitting out exposed to air it becomes ordinary distilled water. Distilled water and deionized water are both fit to drink but deionized water is more pure. This is critical in certain applications such as laboratory experiments. Distillation is one of the oldest methods for creating pure water.
Distilled water conducts electricity whereas deionized water does not. This results in the ph being lower than seven. Distilled water has been boiled so that it evaporates and then re condensed leaving most impurities behind. If you want to test the accuracy of your ph electrodes use ph buffers.
The readings will drift and be essentially meaningless. You can compare the deionized water vs distilled water but to say that the distilled water is the same as deionized water is a mistake. The terms distilled water and deionized water are often misunderstood. As soon as water comes in contact with air co2 gas starts to dissolve it forming carbonic acid h2co3.
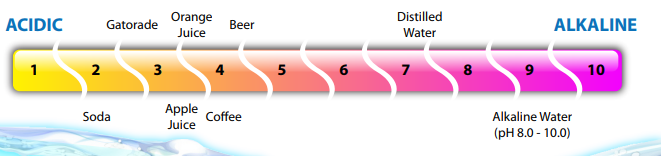
The ph of distilled water is theoretically about 7. Why does the ph of a substance matter. The topic of ph and water has been well documented over the years. The ph scale encompasses 14 orders of m agnitude of the concentration of the hydrogen h ion on a scale of 0 to 14.
You must also keep in mind that water deionised water or distilled water do not have a ph of 7. In practice it s difficult to put an exact number on the ph of distilled water because simple exposure to air can dissolve carbon dioxide.